Plants
PCC plants

The first industrial production of P.C.C. (Precipitated Calcium Carbonate) dates back to the beginning of the eighteenth century in England.
Only in the last thirty years, however, there has been a widespread production of this important by-product of lime following the development of new industrial sectors where it is widely used.
With regard to carbonate micronisation process (G.C.C.), the only factor on which man’s work can act is grain size.
In the P.C.C. production process, the crystallographic formation of the product is also possible.
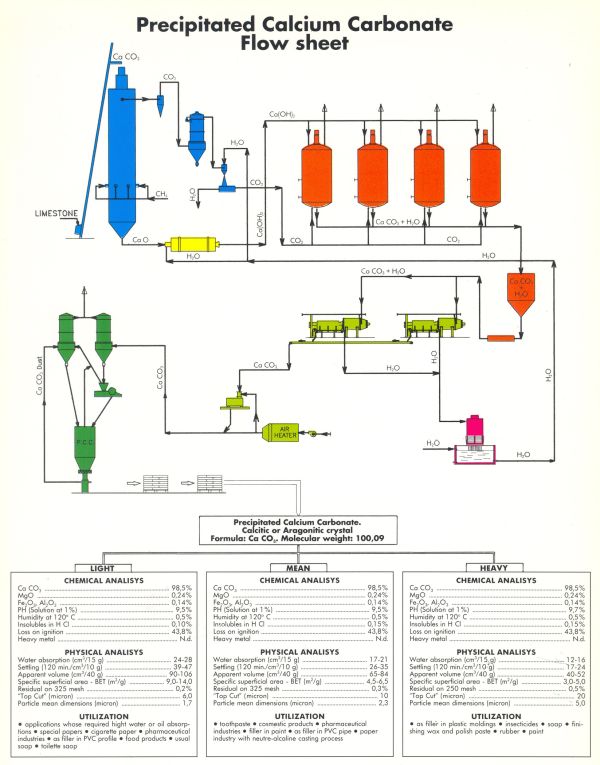
The PCC production process starts with the decarbonisation of limestone (CaCO3) in a kiln, from which lime (CaO) and fumes (CO2) are obtained.
The lime is then transformed into a slurry of lime hydroxide in excess of water (Ca(OH)2 + nH2O ) in an exothermic process, during which it is purified of the heaviest impurities.
The slurry produced and purified in this way is placed in close contact with the CO2-rich fumes coming from the kiln, thus bringing about the recarbonation that leads to the formation of CaCO3 again, which can be used as it is in the form of slurry, for example in paper mills, or dehydrated and dried to obtain a finely dispersed powder.


Acting on the combination of some process variables such as:
- flue gas / lime purification
- lime reactivity
- slaking temperature
- slurry concentration
- CO2 concentration
- process water additive insemination
during the industrial process, it is possible to modify the crystallographic characteristics of the calcium carbonate from the quarry, which makes it possible to vary not only the grain size but also the specific surface area and the degree of whiteness of the product.
SIC, after years of developing technologies related to the production of lime and its by-products with the technological support of two worldwide specialists of P.C.C., started to deal with the design and construction of plants for the production of P.C.C. in 1990.